The classical free-electron model
The classical free-electron model consists of metal atoms arranged in a crystalline structure that can vibrate. This electron has a gas-like perception of electrons. Unlike ordinary gas molecules. This type of electron is so-called because they have free charges.
The velocity of the free electron is 0 and it encounters the absence of the electric field. As soon as an electric field appears, the free electrons move toward the positive potential.
This theory explains why metals can conduct electricity. In this theory more than half of the elements found in the periodic table are metals.
Type of structure:
The free electron model is to show the behavior of valence electrons in a crystal structure of the solid metal. We can find 3 crystal structures:
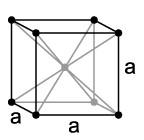
– BCC → Centred cubic lattice, the atoms are located at the vertices in the middle of the cube.

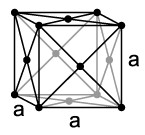
– FCC → Cubic lattice with centered faces, the atoms are located at the vertices and on the faces of the cube.
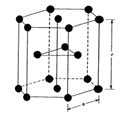
– HCP → Compact hexagonal lattice, unlike the previous ones, this one is located at the vertex of a hexagonal prism, as we can see in the following image:
Tipos de modelos y teorías
In the classical model of free electrons we can find several models that form it:
– Drude’s model → Between impacts, electrons make a free motion with the absence of relating to other electromagnets. The approach of the electromagnets independently is also known as free electrons.
– The Sommerfeld model → In this model we find that the electrons have no relationship to the atomic nuclei, being electrons similar to free particles.
There are 3 free electron theories:
1. Classical free electron theory = this theory was the first in 1900 and the one containing free electrons that are responsible for electrical conductivity.
2. Quantum free-electron theory = building on the previous theory, this theory created in 1928 states that electrons move freely with a constant potential.
3. Zone theory = here we find that the potential is in proportion to three. This theory is known as the band theory of solids.